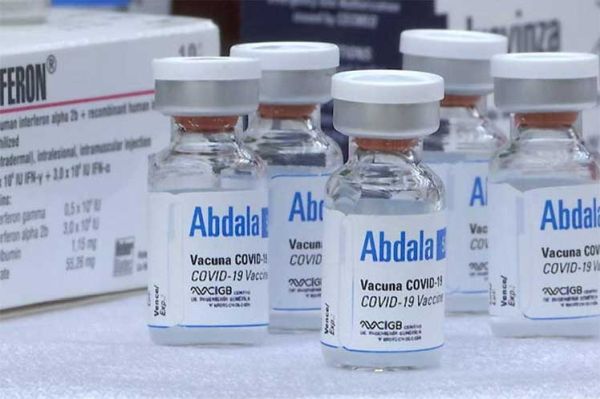
Venezuela will begin production of the Abdala vaccine, one of the five vaccine candidates developed by Cuba against Covid-19, Executive Vice President Delcy Rodríguez informed Thursday.
Accompanied by a delegation of Cuban scientists, the vice-president supervised the Socialist Enterprise’s operations for the Production of Biological Medicines in Caracas to verify the necessary conditions for elaborating the drug.
While the world is engaged in a process of inequality and pettiness in the access of the peoples to vaccines, here is Cuba giving its example and solidarity to Venezuela’, said Rodriguez in statements to the media.
She pointed out that the parties also reviewed all aspects regarding Venezuelan volunteers’ participation in the third phase of Abdala’s clinical trial, which should begin in the next few weeks.
The Vice-Minister ratified the Bolivarian Government’s commitment to guarantee the population’s access to immunization against the SARS-CoV-2 coronavirus, which causes Covid-19, to overcome the pandemic.
In late March, President Nicolás Maduro announced the decision to incorporate, starting in July, the Cuban drug Abdala to the country’s vaccination scheme against Covid-19.
The President reiterated that Venezuela would only admit the use of immunizers approved by the country’s scientific and health authorities, an action aimed at guaranteeing the population’s health.
Maduro also underlined that the country is making progress in the vaccination of prioritized social sectors by using the drugs Sputnik V (Russia) and Sinopharm (China).
The Bolivarian head of state announced the decision not to grant a license for Venezuela’s use of the vaccine developed by the British pharmaceutical company AstraZeneca, due to the complications presented in persons immunized with this antigen.
The Venezuelan authorities informed the World Health Organization that only those drugs approved by the national scientific and pharmacological institutions would enter the country through the Covid-19 Global Access Fund for Vaccines. (COVAX).
 Escambray ENGLISH EDITION
Escambray ENGLISH EDITION





Escambray reserves the right to publish comments.